|
In any atom with two or more electrons, the repulsion between the electrons makes energies of subshells with different values of l differ so that the energy of the orbitals increases within a shell in the order s p > d > f. The energy of atomic orbitals increases as the principal quantum number, n, increases. The specific arrangement of electrons in orbitals of an atom determines many of the chemical properties of that atom. This allows us to determine which orbitals are occupied by electrons in each atom. Having introduced the basics of atomic structure and quantum mechanics, we can use our understanding of quantum numbers to determine how atomic orbitals relate to one another. Relate electron configurations to element classifications in the periodic table.Identify and explain exceptions to predicted electron configurations for atoms and ions.Derive the predicted ground-state electron configurations of atoms.Orbital diagrams: pictorial representations of the electron configuration.Īufbau principle: states that in the ground state of an atom or ion, electrons fill atomic orbitals of the lowest available energy levels before occupying higher levels.By the end of this section, you will be able to: Orbital: physical region or space where the electron can be calculated to be present. Atoms consist of a nucleus surrounded by electrons. Electron configurations and orbital diagrams can be determined by applying the Pauli exclusion principle (no two electrons can have the same set of four quantum numbers) and Hund’s rule (whenever possible, electrons retain unpaired spins in degenerate orbitals).Ītom: the smallest possible amount of matter that still retains its identity as a chemical element.The relative energy of the subshells determines the order in which atomic orbitals are filled (1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, and so on). 
Practice Exam 3 C/P Section Passage 3 Question 15 The orbital diagram of the following elements can be filled as below.Ī core-abbreviated electron configuration (right) replaces the core electrons with the noble gas symbol whose configuration matches the core electron configuration of the other element.Ĭhemistry Question Pack Passage 14 Question 79Ĭhemistry Question Pack Passage 19 Question 106 The orbitals are filled as described by Hund’s rule: having the maximum number of unpaired electrons for the lowest-energy configuration. The remaining two electrons occupy the 2p subshell. Four of them fill the 1s and 2s orbitals. When drawing orbital diagrams, we include empty boxes to depict any unoccupied orbitals in the same subshell that we are filling.Ĭarbon (atomic number 6) has six electrons. There are three degenerate 2p orbitals (ml = −1, 0, +1), and the electron can occupy any one of these p orbitals. Because any s subshell can contain only two electrons, the fifth electron must occupy the next energy level, which will be a 2p orbital. The n = 1 shell is filled with two electrons, and three electrons will occupy the n = 2 shell. The fourth electron of beryllium fills the remaining space in the 2s orbital.Īn atom of boron (atomic number 5) contains five electrons. 
The n = 1 shell is filled in a helium atom, and the electrons are spin paired. We start with a single hydrogen atom (atomic number 1), which consists of one proton and one electron. 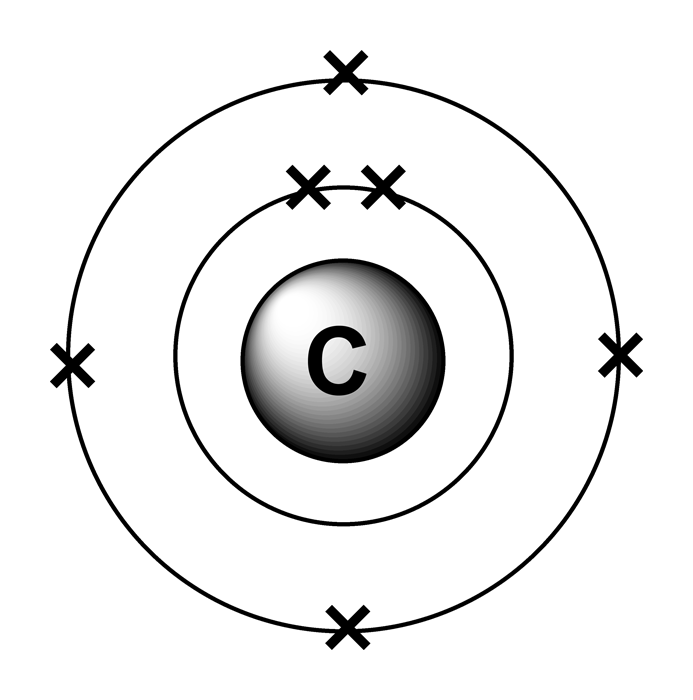
Orbital diagrams are pictorial representations of the electron configuration, showing the individual orbitals and the pairing arrangement of electrons. For example, the 5p orbitals fill immediately after the 4d, and immediately before the 6s. The Aufbau principle, as shown in the figure above, illustrates the traditional way to remember the filling order for atomic orbitals. Electrons enter higher-energy subshells only after lower-energy subshells have been filled. – A superscript number that designates the number of electrons in that particular subshell.Īs the number of electrons in the atoms increased, each added electron occupies the subshell of lowest energy available, with 2 electrons per orbitals. – The letter designates the orbital type s p d or f (the subshell of angular momentum). – The number of the principal quantum shell. We describe an electron configuration with a symbol that contains three pieces of information: The arrangement of electrons in the orbitals of an atom is called the electron configuration of the atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
